Introduction to the Battery University (1/5)

From lead to lithium
There are many types of batteries. The main chemical elements and compounds determine the type of battery. The most well-known are the lead-acid batteries in every vehicle, which start the engine and supply basic consumption when the engine is switched off – see the photo at top right.
However, lead batteries can also be found in trams, trains, and the metro, and in addition to powering the on-board network, they serve as a backup source for brake control when the main power supply is not available.
In these vehicles we can also find other batteries, namely nickel-cadmium (NiCd) or nickel metal hydride batteries (NiMH). NiCd or nickel-cadmium batteries are now produced only for industrial use, due to the content of toxic cadmium. Due to cadmium, they are gradually being replaced by Nickel metal hydride batteries – NiMH. Compared to lead-acid batteries, they have the advantage that they do not suffer when they are insufficiently charged, and they also have a significantly longer service life in industrial variants. Nickel-based batteries have also previously been used as traction batteries for electric propulsion – for example in trolleybuses such as the range extender, ie energy storage for travel without a trolley, or in locomotives. Their main disadvantage is that they are large, and one cell has a low nominal voltage of 1.2 V compared to 2 V for lead batteries.
A major revolution in vehicle propulsion occurs with the advent of lithium batteries. Lithium batteries have a significantly higher energy density, are lighter, smaller, and last more charge-discharge cycles than lead and nickel batteries.
Note: In connection with the propulsion of vehicles, we are talking about so-called traction batteries (from the Latin trahere – pull).
Storage of traction batteries in an electric car
The photos below illustrate the use of lithium batteries in an electric car: The picture above shows a 3D model of an electric vehicle with its traction batteries. The last photo in the article shows the disassembly of the traction batteries of an electric vehicle.

Before we take a closer look at lithium batteries, let’s show one more interesting thing here: lead batteries still power electric boats on the Brno dam. Their disadvantage in the form of heavyweight becomes an advantage here: The heavy batteries at the bottom of the ship also serve as a ballast stabilizing the ship so that it does not overturn.
Different types of batteries - different properties
The lithium battery area itself contains many types that differ in their properties, life and price. Today, there is no universal type that would allow the use of a light, small and cheap battery with a high amount of energy, high performance and longevity, and therefore a different type of lithium battery is suitable for each application.
At Battery School, we will look at each application to understand which batteries are best for them. The graph below shows the specific battery energy in Wh / kg for an initial idea.
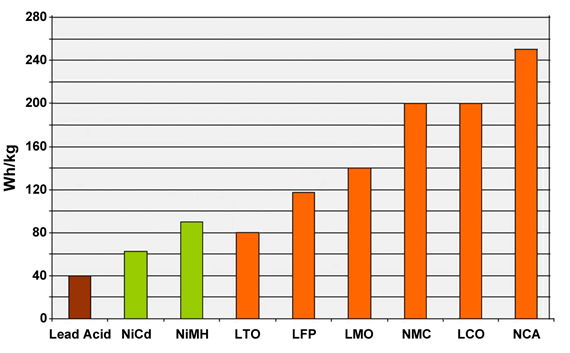
Basic criteria for battery selection
When choosing a battery system for a vehicle, it is then necessary to specify several basic criteria that will help with the selection of a suitable type of traction battery. Especially for public transport vehicles with no standard series production, there is a lot of room for adapting the vehicle to the requirements of a particular carrier.
The first determinant of the type of traction battery is the type of vehicle and its operation.
The battery, which serves as a range extender for partial trolleybuses, will not be suitable for a vehicle with a long range requirement, such as a long-distance electric bus, but may instead be ideal for a fast-charged electric bus at stops, end stations, etc.
On the other hand, a battery that would be used, for example, in an electric garbage truck, will not be entirely suitable for these applications, because it must travel the entire shift without the possibility of recharging.
An important aspect for choosing a suitable technology is also the service life, which results in later costs for battery replacement. Some lithium batteries, such as lithium titanate (LTO), have a large number of cycles and a calendar life and, when properly designed, will allow the same life to be achieved as the planned life of the entire vehicle. Additional batteries will allow all-day operation without recharging and the batteries will be charged when the vehicle is not moving, similar to personal electric cars.
Last but not least safety must also be taken into account. Even in this, the types of lithium batteries differ, so it is necessary to take into account the complexity of the design of the entire battery system.
An afterlife
A separate topic, which we will also address, is the disposal of batteries after they can no longer be used in their original application – the vehicle. Battery disposal and recycling is a burning issue today that is still open. The recycling and recovery of chemical compounds from batteries is still the subject of research by many scientists around the world. Research is not facilitated by the fact that lithium batteries differ in their chemical composition. The research is then based on the possibility of extracting the rare elements that lithium batteries contain, and it must be difficult to mine mostly in developing countries around the world.
Even after the battery has reached the end of its life in the vehicle, because it no longer has sufficient capacity, it can serve as a stationary source of energy. Compared to lead batteries, the degradation of lithium batteries is gradual – simply put, it is linear with respect to style of use and age, on the contrary, lead batteries have a clear calendar life, after which their capacity drops rapidly to zero, and their further use is no longer possible. On the contrary, the recycling of lead-acid batteries is well managed by their manufacturers, and almost all discarded lead-acid batteries can be recycled and thus reused.
The secondary use of discarded lithium batteries, so-called second life, is a new worldwide topic. There are several pilot projects in the world where discarded batteries are used. In our country, for example, nano power uses discarded LTO batteries, which originally operated in AGVs (automatic handling robots), as an energy storage device in its stationary energy storage, which it uses to measure the capacity of new batteries. Here, energy is transferred from one battery to another, making it possible to use old batteries that could no longer be used in the original application and to save costs by significantly reducing the energy consumption required to charge the batteries before capacity measurement.
What does Battery University
Our Battery School will focus on individual applications and analyze which specific technology is suitable for this particular type of application. We will look in detail at partial trolleybuses, electric buses with continuous (opportunistic) and night charging, trams and trains, fuel cell buses and vehicles for city services, such as garbage trucks.

